Towards non-invasive imaging of interfacial water by AFM
Resolving the hydrogen-bonding structure of interfacial water is crucial for understanding many extraordinary physical and chemical properties of water/solid interfaces. To date, scanning probe microscopy (SPM) has been an ideal tool to visualizethe microscopic structureand dynamicsof water at solid surfaces due to high spatial resolution. However, an intrinsic problem of SPM is that all the probes inevitably induce perturbation to the fragile water structure, due to the excitation of the tunneling electrons and the tip-water interaction forces, especially under the close-imaging condition applied in order to achieve ultrahigh spatial resolution. This limitation makes SPM fall short compared with non-invasive spectroscopic methods.
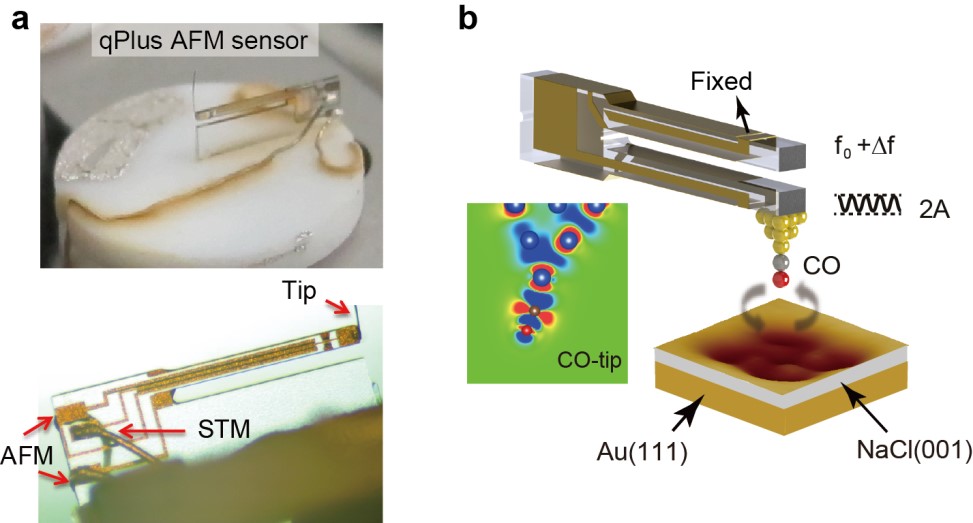
Now, the teams led by Prof. Ying Jiang and Prof. Enge Wang of International Center for Quantum Materials (ICQM) of Peking Universityfind a method to overcome this intractable problem. As published in Nature Communications on 9th Jan. 2018 (doi:10.1038/s41467-017-02635-5), the researchers report the submolecular-resolution imaging of metastable water nanoclusters on a Au-supported NaCl(001) film by probing the high-order electrostatic force using a qPlus-based noncontact atomic force microscopy (AFM) (Fig. 1), which is almost non-invasive to the weakly bonded water clusters.
“At large tip heights where only the long-range van der Waals and electrostatic forces are detectable, the resolutionof AFMis usually quite poorfor weakly polarized molecules.” says Jiang, “However, the situation could be quite different for the strong polar molecules such as water.”
The AFM images of the water tetramers taken with a CO-terminated tip at large tip-water distance show prominent internalfeatures, which resemble the electrostatic potential distribution, whereas, the STM images are featureless. Comparison with the theoretical simulations by the group of Dr. Pavel JelínekfromInstitute of Physics, the Czech Academy of Sciencesreveals that such a high resolution originates from the high-orderelectrostatic force acting between the quadrupole-like CO-tip and the strongly polar water molecules(Fig. 1).
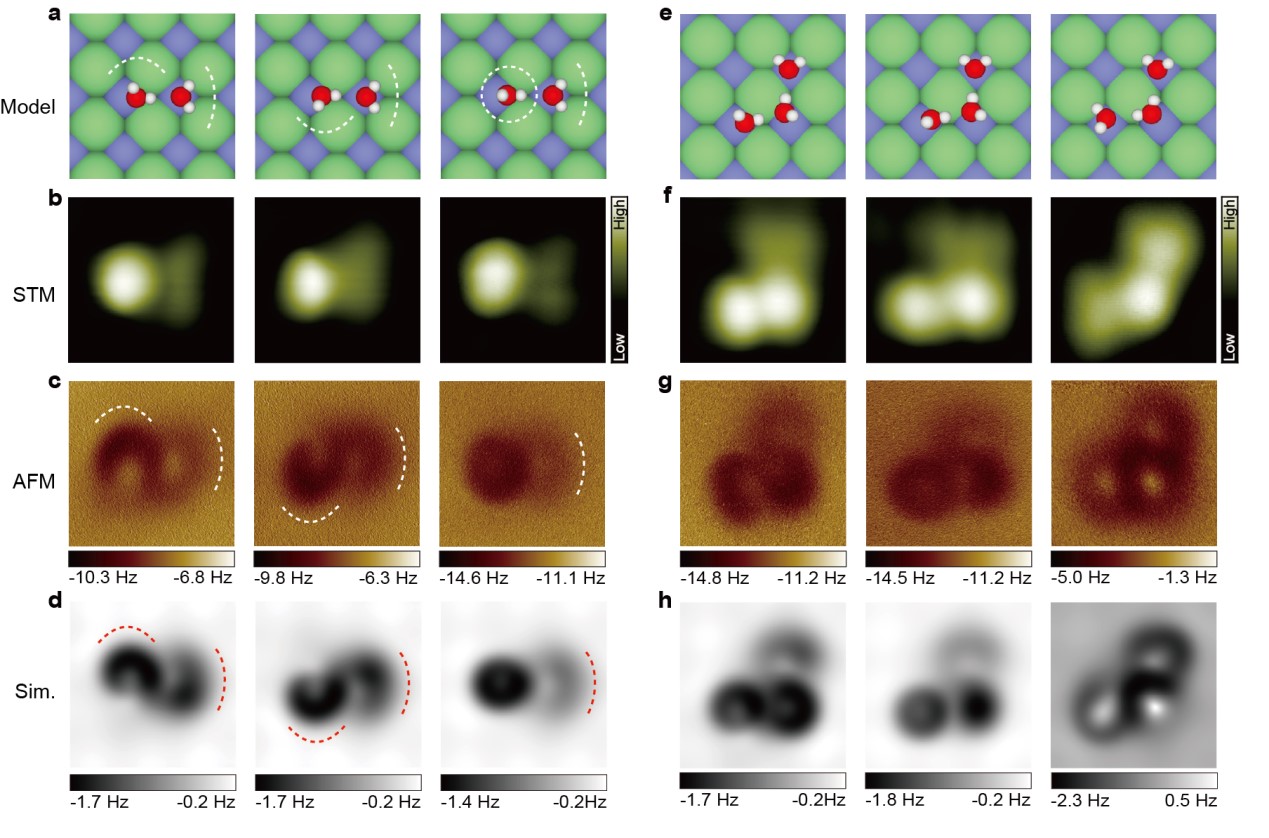
'The magic of the multipole electrostatic forcelies in that on one hand it has short-range character to yield superb submolecular resolution; on the other hand such a weak force signal is still detectable at relatively large tip-water distance, thus avoiding the disturbance of the tip on the water molecules.' adds Jiang. The minimum perturbation of the probes during AFM imaging could be as small as 40~50meV.This technique was found to be very precise for the structural determination of the weakly bonded water clusters and even their metastable states, which have been very difficult to image before with conventional SPM (Fig. 2).
The weakly perturbative imaging achieved in this work defeats the longstanding limitation in the SPM studies of water at surfaces, and may open up a new era of studying the intrinsic or 'hidden' structures of ice/water on surfaces, ion hydration and biological water with atomic precision.The submolecular-resolution AFM images of water obtained by CO-tip not only provide the spatial information of electrostatics, but also allow us to determine the detailed H-bonding structure including the position of the H atoms, which is crucialfor the understanding of H-bonding interaction and dynamics of water.
This work received supports from Ministry of Science and Technology of China, National Natural Science Foundation of China, National Science Fund for Distinguished Young Scholars, Cheung Kong Young Scholar Program, Praemium Academie of the Czech Academy of Sciences, and the Ministry of Education of the Czech Republic.
Article link: Jinbo Peng, Jing Guo, Prokop Hapala, Duanyun Cao, Runze Ma, Bowei Cheng, Limei Xu, Martin Ondráček, Pavel Jelínek*, Enge Wang*, and Ying Jiang*, 'Weakly perturbative imaging of interfacial water with submolecular resolution by atomic force microscopy', Nature CommunicationsDOI: 10.1038/s41467-017-02635-5 (2018).
Figure 1. (a) Experimental setup of a qPlus AFM sensor. (b) Schematic showing the high-orderelectrostatic force acting between the quadrupole-like CO-tip and the strongly polar water molecules.
Figure 2. Non-invasive AFM imaging of metastable water dimers (a-d) and trimers (e-h) with a CO-tip. The crooked depressions in the AFM images arise from the electrostatic potential distribution of positively charged H, reflecting the position of the H atoms.